 
Plug the equation into your scientific calculator. Thus, our answer will be in units of g/L. Mass/V = (16.05 g/mol) x 1 atm / (273 degrees K)īecause of the gas law constant, atm (unit of pressure), K (unit of temperature) and mol (numerical unit) cancel out, and we are left with units of grams per L (unit of volume), which is what we want. Re-arrange the ideal gas law equation, so that you get M/V (which is density). The molar mass and molecular weight is typically given in units of grams per mole.” - ) 
N = mass/ mw (molecular weight- “The molecular weight is essentially the same thing as the molar mass except that, as the name implies, it refers to molecules rather than just elements. Standard Temperature and Pressure is defined as air at 0 C (273.15 K, 32 F) and 1 atm (101.325 kN/m2, 101.325 kPa, 14.7 psia, 0 psig, 29.92 in Hg, 760 torr, 33.95 Ft.H2O, 407.2 In.W.G, 2116.8 Lbs./Sq.Ft.) standard temperature and pressure, standard conditions for measurement of the properties of matter. STP = Pressure (P) = 1 atm (unit of pressure) and Temperature (T) à 273 K (unit of temperature) A Systematic Transfer Planner (STP) allows an investor to transfer a fixed amount at periodical intervals from a liquid fund to an equity fund. Multiply the moles of the limiting reagent by the stoichiometry of carbon dioxide in the reaction to give the moles of CO 2 produced. To find the theoretical yield, you can follow the steps below: Find the moles of the limiting reagent. How to solve the problem: Density is mass over volume. The theoretical yield of CO 2 depends on the reaction taking place and the amount of reagents. Problem: Calculate the density of CH4 gas at STP. fig., but hopefully all my work is accurate.* 
*Remember significant figures! I'm not the best at sig. Helpful reference for checking your findings of molecular weight and molar mass: For example, ΔS° refers to the change in entropy at STP.Confused about how to calculate the density of a gas at STP? Follow these step by step instructions. In this standard formula, the pressure is in torr and temperature is in Kelvin. Standard state conditions, which include standard temperature and pressure, may be recognized in calculations by the superscript circle. You can compute the volume of gas in STP using the standard formula. STP means Standard Temperature and Pressure. How many oxygen atoms are present in 0.12 L of nitrogen dioxide gas at STP 6. If you need to do any calculation regarding this topic feel free to use our Flow. 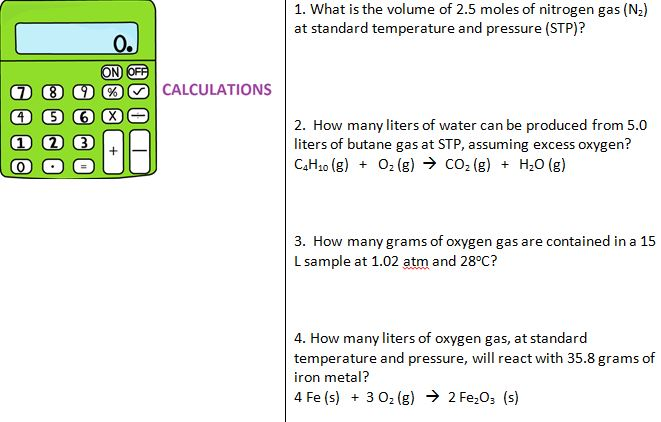
STP commonly is used when standard state conditions are applied to calculations. What mass of carbon dioxide occupies a volume of 1.05 L at RTP 5. Advanced Standard Temperature and Pressure Calculator, Many Unit Input Options, Calculates Gas Volume at. Standard reference conditions are important for expressions of fluid flow rate and the volumes of liquids and gases, which are highly dependent on temperature and pressure.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
